Dugongs

We want you to have a greater ability to learn about some of the unpleasant animal issues in our world.
Furred Animals of Australian
Neville W. Cayley
DUGONG OR SEA-COW
Order SIRENIA (Sea-sirens)
The second group of purely aquatic mammals, known as Manatees and Dugong, have no close relationship with the even more widely sea-faring whales. They are known as Sea-Cows because of their vegetarian diet and mammalian habit of suckling the young, as well as by their somewhat stupid bovine temperament. As in whales, nature replaces the hairy ancestral coat with a thickened hide and under-coat of oily blubber, though fine hairs are discernible about the skin and strong ones are set about the mouth.
Because of ancient belief in sirens who with their songs lured the unwary to destruction or to mansions beneath the waves, the group of Sea-Cows was named Sirenia, although their general appearance has little in common with our modern idea of a siren of the surf. The earliest of such myths may be traced to the Greek and Arab seamen observing the movements of Dugong, while Megasthenes recorded a creature with the appearance of a woman in the ocean near Taprobane (Ceylon). Enlarging upon this, other chroniclers peopled the seas with fishes having heads like lions, probably inspired by the hairs about the Sea-Cow’s mouth, and thus was born the conventional idea of the beauteous blonde mermaid with flowing tresses and fish-like tail, so firmly credited by early Portuguese and Dutch navigators.
As early as 1816 a great naturalist separated the Sirenian order from that of the whales, and later associated their origin with the elephant section of the hoofed order of mammals. This theory of relationship is maintained by the formation of the face which suggests a gradual retraction of a trunk-like proboscis, and by the fact that in most Sea-Cows and all elephants there is a unique provision for the replacement of worn dentures by the continual growth and sliding forward of the molars in a way that would be a priceless human asset. The suckling of the young between the fore-limbs is another anatomical similarity as the teats of the elephant are also placed between the fore- quarters.
Living representatives of the many extinct forebears of the Sirenia include the African and American Manatees, the nailless Manatee of the Amazon and Orinoco, and the Dugong of the Red Sea and certain parts of the Indian and Pacific oceans. Never a large family even in early geological times, the Sirenia are now sadly depleted. One of the most distinguished members, zoologically speaking, the giant Steller’s Sea-Cow, discovered by the navigator Behring in 1741, became extinct within thirty years of its discovery by man. The American Manatee was threatened with a similar if slightly retarded fate until the Government, realizing its scientific interest and popular attraction, passed protective laws involving a penalty of five hundred dollars, an effective form of protection which should certainly be applied to our local Dugong, because of its ever shrinking coastal range and population.
Distribution. — Once occurring as far south as Botany Bay, Sydney, but only traceable on the north coast of New South Wales by the bones of past aboriginal feasts. Now occurring sparsely about Moreton Bay where they once gathered in herds of several hundred. Northward the range continues within the Great Barrier Reef and around Torres Strait and across to Broome in Western Australia, formerly southward at least to Sharks Bay, as well as about adjacent islands eastward to the Solomons.
Notes.— The popular and generic names of this submarine elephant are both derived from the Malayan “duyong” and Javanese “duyung.” Like all Sirenian mammals they are also known as Sea-Cows because of their somewhat bovine ways and habit of browsing upon submerged aquatic plant life. The bones are noted for their heavy density, especially the skull and ribs, the weight of which helps, without effort, to keep the ungainly animals below when grazing along their submerged pastures.
In small herds they frequent shallow seas, bays, and estuaries, and are inoffensive and sluggish creatures, except when alarmed, and are apparently endowed with comparatively small intelligence. Usually a single calf is born, and is tended with assiduous care by the mother who holds the nursling partly clasped to the breast when rising for air or to suckle it. The adult length ranges from seven to nine feet, occasionally reaching nine feet six inches. The males have slightly projecting ivory tusks analogous with those of the ancestral elephant stock; the tusks are usually worn down by sea-grit, but do not pierce the gums in females. The colour of the skin (usually scarred by contact with jagged coral, and possibly the tusks of males) varies from reddish-brown to bluish or greenish olive-grey with fleshy-white under-parts.
When feeding, their flexible flippers are used in moving about and, possibly, as in the American Manatee, to help convey food towards the mouth. The remarkably mobile and enlarged upper lip is the main aid in feeding as it is sufficiently prehensile to grasp the Zostera-like marine grasses and draw the food into the mouth, aided by the lip-bristles. When the writer and a colleague, A. A. Livingstone, were collecting for the Australian Museum in the Santa Cruz Group in 1926, our host, N. S. Heffernan, District Officer at the time, stated that Dugong were fairly common there and in the Solomons. He shared the view of the natives that they also rooted in the sand for various kinds of shell-fish, thus explaining for the wearing of the tusks of the males, and that the female relies upon gobbling pig-like with her tough snout. It was also believed that only the lily-like roots of the dugong-grass were eaten, thus accounting for the floating grass used as a sign by aboriginal hunters.
Of the herd instinct and love for their young and the companionship of their fellows Banfield, in Confessions of a Beachcomber, wrote of them about his beloved Dunk Island as “Rolling and lurching along gambolling like good-humoured, contented children” as the herd leisurely moves to and from the favoured feeding-grounds; occasionally sending up fountains of spray by splashing mightily with their powerful tails, like “great unreflecting, sportful water-babes.” He also wrote that they seemed to have an affection for each other which was almost humanly demonstrative. When one of a pair was killed the bereaved lingered about the tragic spot for days, and when rising to the surface its gulping sighs sounded like expressions of the keenest grief. The tender-hearted “Beachcomber” considered it possibly pure fancy, but it affected him so strongly that he discouraged the blacks as far as possible from killing them.
Adult Dugong, like whales, have no definite voice, so that if the sirens’ singing was based on that of the Sea-Cow, it must have represented a highly imaginative conception of the whistling sigh of air rushing through the nostrils — although Dugong calves are said to have a bleating cry like a young lamb. In his Creatures of the Sea Bullen quotes instances of the taming of Sirenians as an indication of their once higher land intelligence. As when a retired Superintendent of the London Zoo cared for a young Manatee in Surinam, which came to him as he waded into its pond and was held between his knees while feeding from a bottle. Another, kept by a Spanish American Governor for twenty-six years, is said to have behaved as sensibly as any land mammal and to have allowed the boys of the household to ride on it about the lake.
The aborigines’ method of hunting (used long before the advent of whites) is by means of an ingenious harpoon-spear in which a barbed dart, attached to a rope, is jammed into a socket at the end of the shaft. The black hunters either hurl them from platforms built above grazing-grounds or scout in frail bark canoes for signs of floating grass, watching silently and striking as the browsing Dugong rises to breathe. The first aim is the tail as the animal may then become entangled in the line; a great rush usually follows with the canoe bouncing wildly as the hunters steer with bark-paddles until the prey weakens and another spear is thrust through the nostrils to suffocate it.
About the year 1860 a medical officer in Queensland lectured upon the curative properties of Dugong oil in lung complaints. This led to a popular belief that a valuable remedy for consumption had been found; a leading firm of chemists actually ordered a thousand gallons at three guineas a gallon. It is doubtful, however, that the oil has properties much superior to many other animal fats and oils. By 1895 the price fortunately fell as low as about 12s. a gallon, with correspondingly reduced incentive for slaughter.
The main method of capture by whites was with yard-mesh coir nets. About fifty years ago fishing was conducted with a schooner as a floating station, and small boats for netting; but the venture was not successful, doubtless because of the rapid reduction of quarry due to destruction and disturbance of the slow-breeding creatures in their favoured haunts.
It is estimated that a fully grown animal yields up to six gallons of oil from its fat. The hide, nearly an inch thick, makes very good leather when properly treated; when cut up green and boiled it is convertible into glue. The ivory tusks of males, about nine inches long, make fine carver handles, while the bones are said to provide the best charcoal for sugar-refining. They were hunted by early settlers for the flesh which is said to resemble pork or veal and to be nutritious and tasty, and an excellent substitute for bacon when rolled and smoked.
The hunting for food by aborigines is natural and hardly constitutes a vital menace. But their employment as hunters for commercial gain by Europeans, who also use large nets at times, would inevitably end in banishing these intriguing creatures from our shores. In 1922 Banfield deplored the increasing rareness of Dugong, which he attributed mainly to freebooting trochus- and trepang-fishing vessels. He wrote that Japanese luggers occasionally appeared at Dunk Island, when “generally within a few minutes dinghies were launched for a Dugong hunt, as the crews are extremely fond of the meat and lose no opportunity of harpooning the harmless creatures.”
As Bullen said, they are far too gentle for the stern world of to-day; as much out of place in it as a herd of antelope in a jungle inhabited by tigers. It cannot be over emphasized that their slow rate of breeding and defenceless ways must inevitably lead to extinction if slaughter by modern means is permitted. Any protection at present afforded should be permanently and rigorously enforced. Not only are Dugong amongst the least harmful creatures of the sea, but their elephantine descent renders them of remarkable scientific interest; while, as the age-old inspiration of the mariners’ mermaid myth, their presence along the Great Barrier Reef must ever add to the tourist attractions of that marine wonderland.
Neville W. Cayley, Furred Animals of Australia (New York: C. Scribner’s Sons, 1947), pp. 240-244.
See: https://babel.hathitrust.org/cgi/pt?id=coo.31924002882912&seq=1&q1=
The Animals of Australia
A.H.S. Lucas and W.H. Dudley Le Souef
The Dugong
Halicore dugong.
Skin, thick and smooth, with a few scattered hairs. Upper lip large, thick and bristly. Flippers, short, thick and fleshy. Slaty or brownish-black above, whitish below. Length, 8 feet. Shores of the Indian Ocean, Malay Archipelago, and Northern Australia, on the East as far south as Moreton Bay.
Ungainly creatures with small brains and very limited intelligence, hence too easily captured or destroyed, and now greatly thinned in numbers. In some cases the flesh is eaten, but the chief value lies in the oil, which is obtained from the layer of blubber beneath the skin, and which is very highly thought of, being free from odour and a good substitute for cod- liver oil. The Dugong feeds entirely on sea-weeds and marine phanerogams. It does not leave the water to browse on the land herbage, as it is quite unadapted for travelling on the land, having no hind limbs and very weak fore-limbs.The Dugong is the original of the Mermaid. The females are gentle and affectionate to their young, and have the habit of sitting up in the water to suckle them, holding the calf to the breast by the flipper, with an almost human attitude and expression. Hence the stories, magnified in the forecastle, of the beautiful maiden with the fish’s tail.
The following interesting account is given by Mr. A. H. E. Mattingley in the Victorian Naturalist: —
“One of the methods adopted by the natives for their capture is when they discover where the Dugongs come to browse on the sea-grass, which grows on the mud-banks near the shore, to erect a staging on which to stand, and then on moonlight nights to take up their position on the staging with a harpoon and coil of rope. The harpoon consists of a long pole with a hollow in one end, into which is fitted a wooden head, which is attached to the middle of a pole by a grass rope. Upon espying a Dugong they plunge the harpoon into it, whereupon the animal immediately rushes off; the harpoon head becomes dislodged from the pole, but being tied to the pole at its centre, retards very effectively the animal’s progress through the water. The native paddles after it in his canoe, waiting till the animal becomes exhausted before finally despatching it.
“Another method is to spear them from a canoe in the day-time as they are making their way to some feeding ground. The spear used for this purpose is composed of a light wooden shaft in which is imbedded a piece of sharpened fencing wire. This is easily plunged through their gutta-percha like hide, and, as the animal dashes off, the wire bends like a fish-hook, and tows the shaft or some other float behind, and is easily followed by the natives in the canoe. The spear thrust is, however, not sufficient to fatally injure the animal, and it is therefore necessary for the natives to kill it, which is done by suffocation. Being a mammal, it is necessary for the Dugong to come to the surface to breathe. One of the natives dives overboard, and endeavours to insert a wooden plug into the animal’s nostrils, and so cause suffocation; failing this, they usually tie a rope round its tail, and drag it down under the surface of the water, and so drown it; in the event of both these methods failing, they spear it through the nostrils.”
The white man’s method is equally cruel, and as the cows are killed off in large numbers for the sake of the oil, unless adequate protection is enforced, it can only be a matter of time before the Dugong will be exterminated in Queensland. Bones of the Dugong were met with during the excavation of Shea’s Creek, which runs into Botany Bay. Hence the animal must formerly have extended much further south than at present.
A.H.S. Lucas and W.H. Dudley Le Souef, The Animals of Austraia; Mammals, Reptiles and Amphibians (Melbourne: Whitcombe and Tombs, Ltd., 1909), pp. 1-62
See: https://babel.hathitrust.org/cgi/pt?id=mdp.39015082368757&seq=1&q1=
Extinct and Vanishing Mammals of the Western Hemisphere
Glover M. Allen
Order SIRENIA: Sea-cows
The sea-cows are completely aquatic in habit and somewhat fishlike in external form. They are thought to be distant relatives of the elephants. Three recent families are recognized, each with a single genus:
(1) Dugongidae, the dugong. This sirenian occurs in the Red Sea, along the coasts of East Africa, Ceylon, the islands of the Indian Ocean, the Malayan Archipelago, the Philippines, and northern Australia. It is in need of protection.
(2) Hydrodamalidae, Steller’s sea-cow. The single species had no teeth and was restricted to two small islands in the northern Pacific. It was exterminated by the Russian hunters.
(3) Trichechidae, the manatees. Three species (the Florida manatee is considered a race of the West Indian species) are still extant. Manatees occur in tropical and subtropical America and the west coast of Africa. — J. E. H.
Family DUGONGIDAE: Dugongs
DUGONG
DUGONG DUGON (P. L. S. Müller)
Trichecus dugon P. L. S. Müller, Linnaeus’s Vollständigen Natursystems, Suppl., p. 21, 1776 (Cape of Good Hope to Philippines).
SYNONYMS: Halicore hemprichii Ehrenberg, in Hemprich and Ehrenberg, Symbolae Physicae, Mamm., dec. 2, folio k, footnote, 1832 (Barkan Island, Red Sea); Halicore lottum Hemprich and Ehrenberg, Symbolae Physicae, Mamm., dec. 2, folio k, footnote, 1832 (Hauakel Island, Red Sea); Halicore tabernaculi Rüppell, Mus. Senckenbergianum, vol. 1, p. 113, pl. 6, 1834 (Red Sea); Halicore indicus Desmarest, Encycl. Méth., Mammalogie, pt. 2, p. 509, 1822.
AUSTRALIAN DUGONG
DUGONG AUSTRALIS (Owen)
Halicore australis Owen, in Jukes’ Narr. Voyage Fly, vol. 2, p. 323, 1847 (Endeavour Strait, North Australia).
FIGS.: Prater, 1928, pls. 1-4; Dollman, 1933, figs. 1-6 (exterior and skull).
The dugong is found in warm tropical and subtropical coastal waters from eastern Africa to the Philippines and Formosa, as well as southward to the east coast of Australia, and eastward again to the Solomon Islands. Although separate names have been given to those from the Red Sea and from Australian waters, it is not at all certain whether these represent distinguishable forms or what the characters are if they deserve recognition. Dollman (1933) found on comparing skulls from the East African coast with others of about the same age from Australia that the former were “much less massive and smaller” than the latter. In view of the present uncertainty the supposed forms may be considered together. On account of the palatability of the flesh this animal is much hunted wherever found and hence is in danger of reduction.
The dugong is in external form much like a cetacean, spindle-shaped in body, with the tail ending in lateral flukes, which are notched in the median line and pointed at the outer end. The skin is of a blue-gray color, lighter below. The fore limbs are flipperlike, but the hind limbs have disappeared in modern forms; eyes are small, the ear-opening a minute pore. The muzzle forms a flat disk, with a vertical cleft in the center, and with short spines on its lower part, while a few short bristles are present on the upper part. The nostrils open separately at the summit of the snout. The skull is remarkable for the downward bend of the rostrum, which in the male has a pair of short tusks, the second pair of upper incisors. The cheek teeth are six in number, but with age the smaller ones drop out in front, and the posterior ones wear down, until there may be but a single one left, the last in the series. Length, up to 10 feet.
Although the type locality of the dugong as given in Müller’s original description is Cape of Good Hope to the Philippines, the former region is probably to be taken only in a general sense; at all events at the present day it is not apparently found farther south on the East African coast than Delagoa Bay, Mozambique (latitude 26° S.), where Barrett (1935) mentions seeing several adults and a “calf” caught in fish traps by the natives, in 1908-10. It is occasionally taken also in fish nets on the coasts of Madagascar and the neighboring small islands, and on the Tanganyika coast. According to Kaudern (1915) it was more often hunted formerly in Madagascar than now, though occasionally seen offered in the markets at Analalava; Petit shows that 20 years ago it was fairly common, but now is much more restricted among these islands. On the coast near Lamu, Kenya Colony, Arthur Loveridge (in G. M. Allen and B. Lawrence, 1936, p. 125) says that according to native report it is still fairly common, and one was brought to him there in 1934; but although the meat has a high commercial value, its captors refused to let him have the skull for $2.50!
Anderson and De Winton (1902), quoting a paper by Krauss, of 1870, on specimens collected by Klunzinger at Kosseir near the head of the Red Sea, speak of the dugong as then “apparently plentiful,” going in small groups of two to ten. They appeared annually on the Nubian coast, especially near Aesa, and in December and January moved northward at least to the island of Safadje. They are little seen by day but appear to be more active, probably feeding inshore, by night.
The single young one is born in winter, and the period of gestation seems to be about a year. Maj. S. S. Flower, writing in 1932, says that it is “now rare on the [Red Sea] coast of Egypt,” seldom coming farther north than latitude 25° N., but to the southward it was not uncommon. The Arab fishermen prize the teeth highly. De Beaux (1931) writes that it is captured off Raheita, southeast of Assab, more frequently than elsewhere in the nets of the fishermen; the meat is said to be tender and well flavored and is much esteemed locally. He strongly urges its protection in the Red Sea and Somali coastal waters. It is an interesting fact that since the opening of the Suez Canal a female of this species was reported by Aharoni (1930, p. 330) as having been killed by fishermen from Tantura, about halfway up on the coast of Palestine, in a shore cave; but the possibility of this having been a monk seal should not be overlooked.
Passing eastward, Blanford (1876) writes that although reported by Murray as occurring on the Persian coast, he knew of no certain record of its presence on the west coast of India farther north than Canara, nor did he know of it from any part of the Arabian coast east of Aden. It is, however, still found on the coasts of Ceylon. In Tennent’s time (before 1863) it was apparently common “from the bay of Calpentyn to Adam’s Bridge,” and about the Gulf of Manaar, but W. W. A. Phillips (1929) implies that it is no longer found in any numbers there and “appears to be in danger of extermination, if not protected.” Millett (1914), however, speaks of it as “plentiful in and about the Gulf of Manaar It is frequently caught in the large shallow lagoons near Trincomalie, where it enters the narrow mouths at high water in search of a certain kind of food of which it is very fond, and where it gets stranded by the receding tide, when it is easily captured.” A more recent note from A. C. Tutein Nolthenius (October, 1936) states that small numbers may still be found in the shallow seas off the northwest coasts of Ceylon, to the north and northeast coasts.
Fifty years ago it appears to have been much more common and somewhat more widely distributed than today. Its flesh is eagerly eaten by certain castes of the indigenous population and is in special demand because of its reputed aphrodisiacal properties. The animal is therefore taken whenever opportunity offers. Usually it is caught in nets spread among the coral reefs, to the vicinity of which it resorts during the northeast monsoon. At one time a considerable number were sent by train, alive, to Colombo, for sale in the fish market, but the practice has been stopped in recent years on the score of cruelty. The dugong is in no way protected at present, and its numbers are becoming much depleted. Unless given some measure of protection very soon its extermination in Ceylon waters may be accomplished at no very distant date. Prater (1928), a decade ago, wrote that the dugong was becoming increasingly scarce in Indian coastal waters and that recommendations were about to be put forward for its protection during part of the year.
Eastward, the dugong occurs in small numbers in the Straits of Malacca, on the Siamese coasts, on the coasts of Sumatra and Borneo, eastward to the Philippines, and thence follows the Japanese current northward even to Formosa and the Riu Kiu Islands, or farther. According to Ridley (1895, p. 165) it was “tolerably common in the Strait between Johore and Singapore; but one does not often see it. However, the Chinese sometimes catch it in nets when fishing, and sell it in the markets as food.”
In the Philippines it “occurs along all coasts from the Batan Islands at the northern extremity” of the group “to the Sibutu group east of the northern part of Borneo. It is not abundant anywhere, but is well known to all fishermen. It is harpooned or captured in fish corrals, but is taken more accidentally than otherwise” (Herre, 1928, p. 1072).
While there are no recent records for Chinese waters, it very likely was once found on the southern coasts, for as lately as 1931 one was captured near Tai-jyu-bo, on the west coast of Formosa and its skull secured by Hirasaka (1932). This author also adduces some interesting information on the former abundance of the dugong in these seas. He adds the report of two others taken the same year at Haikau, a port near Koshun, and states further that many decades ago dugongs were “fairly abundant in the water[s] surrounding the Ryukiu Islands and Amami-oshima. . . and were hunted eagerly for flesh and oil. This hunting was carried on regularly once a year by feudal lords for tribute in Aragusuku Island, near Ishigakishima . . I have heard from an old fisherman that once one was caught in a large trap seine for yellow-tails, off Abratsu, a port on the eastern shore of Kyushu about thirty years ago.” The present condition of these animals, he states, “is rather deplorable” as they are “on the point of extinction and are extremely rare” in these waters. However, the Government of Formosa, on advice of the Committee for the Preservation of Natural Monuments, “intends to forbid the capture of this animal or the disturbance of its resorts.”
While the dugong seems to be mainly prized for its flesh, which is said to resemble pork or bacon (or “between beef and pork”) and for its oil, hide, and even tusks, we learn from an old work published in 1665, concerning the famous mission of de Goyer and de Keyser from Batavia to Canton and Peking, that the Chinese held in high esteem certain stones found in the heads of these sea-cows, which they say have the property of clearing the kidneys of every kind of sand and gravel and of removing obstructions from the lower parts afflicted by them. These “stones” were very likely the dense tympanic bones of the dugong. Sowerby, in discussing this matter, states that so far as known to him there is no record in Chinese literature of the dugong’s occurrence in the coastal waters of that country.
Whether the Australian dugong is identical with that of the East Indian and Formosan waters is still uncertain. It formerly ranged southward on the east coast to at least Moreton Bay, and probably still farther, for skeletal remains have been found at Botany Bay, New South Wales. It is common along the northern coast in tropical waters and occurs on the west coast at the present day, as far at least as Sharks Bay. Somewhat north of this point, in the Kimberly district, Dampier, in his account of 1688, found it in plenty, and relates that “our strikers brought home Turtle and Manatee [i. e., dugong] every day, which was our constant food” (Troughton, 1932, p. 176). Various recent writers agree that on the northern, western, and eastern coasts of Australia it is now getting scarcer owing to continued persecution.
In his excellent account of the dugong, Troughton (1928, 1932) says that in 1893 large herds were a common sight in Moreton Bay, Brisbane, and fishing or netting was carried out there and at Hervey Bay, but the slow breeding of the animals has usually put a stop to ungoverned slaughter. They are not so plentiful now between Brisbane and Cairns, eastern Queensland, and range round the north coast of Australia to Broome on the west coast. In July, 1893, a herd in Moreton Bay was reported as extending over a length of about 3 miles with a width of 300 yards.
The main method of capture by whites was with strong coir nets of a yard mesh, which are not usually more than 300 feet long and 25 meshes deep, anchored at the ends and buoyed with floats. About 50 years ago (ca. 1890), fishing was conducted with the use of a schooner as a floating station and smaller boats for netting, but the venture proved unsuccessful, probably because of the rapid diminution of the quarry.
In 1922 Banfield deplored the increasing rarity of dugong and attributed this largely to their slaughter by the crews of Japanese vessels fishing in the waters of northern Australia for trochus and trepang. They depended for meat on the dugong, which they harpoon from small boats. Pearling crews also kill many for food. In this destruction the aborigines also play a certain part, but though this, Troughton believes, “may hardly constitute a vital menace,” their employment by Europeans in hunting dugongs may do so. He especially points out that “their increasing scarceness, defencelessness, and slow rate of breeding render it essential that the protection at present afforded them should be extended for all time and rigorously enforced.”
The dugong is presumably altogether a vegetarian, browsing on certain marine plants that grow in shallow depths. In eastern Australian waters, stomach examinations by Drexler disclosed two species identified as a Halophila and a species of eel-grass (Posidonia). Hirasaka (1932) states that the stomach of the one reported killed in Formosa in 1931 was said to contain “quantities of marine algae and crabs,” but the account was obtained at secondhand and one may doubt if the crabs were part of the diet. Perhaps other foods will be found to be eaten. Such knowledge is essential to any intelligent reservation of certain waters as sanctuaries for them.
Troughton (1928) summarizes the use made of the dugong in past years in Australia. “They are hunted for their flesh, which is both nutritious and tasty, and, when rolled and smoked can be converted into a substitute for bacon for which there was a ready sale some years ago. A more important product is the oil obtained from the blubber; this is cut into cubes and boiled in water from which the oil is skimmed and refined for medicinal use in the treatment of lung complaints and rheumatic troubles. It is estimated that a fully grown female in good condition will yield sufficient fat to supply from five to six gallons of oil. The hide, which is nearly an inch thick, makes very good leather when well tanned, and when cut up green and boiled can be converted into glue. Tusks attain a length of nine inches, and when polished make handsome carver handles, while the bones are said to provide the best charcoal for sugar-refining.”
As long ago at least as 1863, the oil was believed to be of excellent medicinal value and was in considerable demand in lung troubles. Gould (1863, vol. 1, p. xxxix) wrote that it was “preferred to cod-liver oil, as being less disagreeable to the palate and more easily retained in the stomach.” It is doubtful, however, if its properties are in any way superior to many other animal fats and oils.
There can be little doubt, writes Dollman (1933), that dugongs are in need of protection at the present day since they are being killed off wholesale for food purposes. especially true of those around the coast of Tanganyika Territory and Mafia Island, in the Red Sea, and probably Northern Australia.
Glover M. Allen, Extinct and Vanishing Mammals of the Western Hemisphere with the Marine Species of All the Oceans (New York: American Committee for International Wild Life Protection, 1942), pp. 528-535
See: https://babel.hathitrust.org/cgi/pt?id=mdp.39015023915989&seq=1&q1=
A REVIEW OF THE LITERATURE OF THE DUGONG (DUGONG DUGON)
Sandra L. Husar
National Fish and Wildlife Laboratory
U.S. National Museum Washington, D.C. 20560
INTRODUCTION
The dugong (Dugong dugon) is a member of the family Dugongidae in the order Sirenia. Sirenians, or sea cows, have an extensive fossil history and appear to have descended from terrestrial herbivores before the Eocene. They are thought to be most closely related to the elephants. The huge (up to 7.5 m in length) Steller’s Sea Cow (Hydrodamalis gigas), a close relative of the dugong, was exterminated as late as 1768, within 30 years after its discovery in the Bering Islands. Today, the only remaining species are the Indo-Pacific dugong and three species of manatees, which are found along the Atlantic coasts and in adjacent rivers. All are large, nearly hairless, spindle- shaped aquatic mammals distributed within equatorial latitudes. This group was named for the sirens of ancient mythology. Early mariners returned with reports of strange mermaids, and the combination of hairless bodies, pectoral teats, and shy habits probably guided their imaginations toward this conclusion.
The dugong is listed as vulnerable in the International Union for the Conservation of Nature Red Data Book. Although no accurate population estimates have been made, it is generally concluded that numbers are reduced and declining in most areas. Data on the biology and life habits of these animals are few, and basic information is necessary before sound conservation measures can be taken. . . .
DISTRIBUTION, ABUNDANCE, AND TRENDS
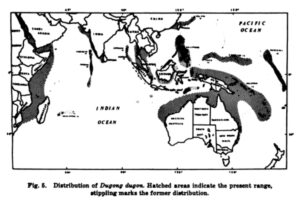
Africa: In Africa the range of the dugong (Fig. 5) [See below.] extends from Durban, South Africa (Best 1968), north along the coast to Egypt in the Red Sea (Stoddart 1972; Gohar 1972; Gohar 1957). Although this seems to encompass a substantial area, the distribution is discontinuous. Populations are found in isolated channels and bays and some local herds are known to have been extirpated. Dugongs were abundant in Madagascar (Malagasy Republic) in 1908 (Prater 1929a), and in the islands around Analava (on the northwest coast of Madagascar) they were fairly common as late as 1922 (Allen 1942); however, only a small population remains today (Philip and Fisher 1970).
In 1691, great numbers were reported from east of Madagascar in the Mascarene Islands by Leugat (in Blyth 1859). Herds of 300-400 animals were sometimes hauled out and shot, and Blyth (1859) believed that they were exterminated on these islands at the time of his writing. An aerial survey carried out on the African side of the Mozambique Channel (Hughes and Oxley-Oxland 1971) showed one of the areas of greatest dugong concentrations to be in the vicinity of Antonio Enes. A substantial population exists here; however, the status of the dugong else- where in Mozambique is unknown.
According to Kingdon (1971) and Dollman (1933), the Pema-Zanzibar Channel and the Rufigi-Mafia Islands off the coast of Tanzania may support stable populations. Dugongs appear to be more abundant in Kenya and the Somali Republic than elsewhere along the coast of Africa (Philip and Fisher 1970), with the Kiunga Archipelago and the Lamu Inland Sea supporting the major populations (Kingdon 1971; Jarman 1966). According to fishermen in this region, there has been no decline in numbers in the area, although they did believe populations to be decreasing outside of the inland sea (Jarman 1966). Dugongs were commonly found in or near Chiamboni, Formosa Bay, and Malindi in the past, but today they are rare (Jarman 1966). The population in the Lamu area apparently maintains itself despite hunting pressure. Ngomeni, between Kipini and Malindi, is also reported to have a stable population. Although Malindi presumably supports many dugongs, their occurrence there is sporadic (Jarman 1966). Large herds have been sighted at both Mombasa and Ma- lindi, but in 1961, numbers in these iso- lated populations appeared to be low (Jarman 1966).
Off the Republic of Somalia, Travis (1967) observed herds of up to 500 individuals and dugongs can still be found in the shelter of Ras Bur Gao and along the archipelago to Kisimayu, on the the equator (Jarman 1966). Fishermen once reportedly observed them along numerous creeks south of Ngomeni, but they are now absent. Some individuals wander as far north as Brava, where they are more common during the rainy season (Jarman 1966). Records are occasional and sporadic from the Gulf of Aden (Funaioli and Simonetta 1966).
In general, dugongs seem to be less endangered along the east coast of Africa, especially in Kenya and the Somali Republic, than elsewhere in their range except perhaps Australia (Fitter 1968).
Anderson and De Winton (1902) reported dugongs as still plentiful in the Red Sea, appearing annually on the Nubian coast near Aesa, and as far north as Safadje. Today, few remain in the Red Sea (Bertram and Bertram 1966a and 1973; Norris 1960; E. Clark, personal communication). They are also extremely rare in the Gulf of Aqaba, the Gulf of Suez, and the Persian Gulf (Bertram and Bertram 1973; Gohar 1957) and are not found off the island of Socotra (Bertram and Bertram 1973).
India-Ceylon (Sri Lanka) Region: Snow (1970) ascertained that dugongs no longer occur in the Laccadive and Maldive Islands or along the Chagos Archipelago. A recent survey conducted between January and March 1970, indicates that dugongs are becoming increasingly rare in Ceylonese waters (Bertram and Bertram 1970a and 1970b), where previously they were common along the coast of Ceylon from the Bay of Calpentyn, north along Mannar, up to the extreme northeast tip of the Jaffna Peninsula (Jonklass 1960; Prater 1929a; Phillips 1927). The range is now restricted to northwest of the island from Jaffna to Puttalam (Bertram and Bertram 1970a and 1970b; Deraniyagala 1965).
At one time large dugong herds of several hundred existed in the Gulf of Mannar, which separates India from Ceylon (Prater 1929a). They were abundant enough to support a large commercial fishery operating from the Ceylon coast; however, the resultant decrease in the population terminated this enterprise (Prater 1929a). From 1957 to 1959, 100-150 dugongs were taken yearly in the Mannar district by individual fishermen, but numbers caught in Ceylonese waters have dropped remarkably in the last 10 yr [years] (Bertram and Bertram 1970a and 1970b).
Prater (1929a) first reported a decline in Indian waters some 40 yr [years] ago. The northernmost occurrence of dugongs in India appears to be off Piroton Island in the Gulf of Cutch (Lal Mohan 1963; Jones 1960). Though once reported from the Malabar Coast (Gibson-Hill 1950), dugongs no longer occur there (Jones 1967a). In eastern India, dugongs range from south of Madras to Cape Comorin (Jonklass 1960).
Overall, abundance in Indian and Ceylonese waters appears to be drastically reduced, with remnants of the population remaining in the region near the Mannar Peninsula, from Jaffna to Puttalam. This scarcity is attributed to the marked increase in local marine fishery (Bertram and Bertram 1970a and 1970b).
Isolated Islands: Snow (1970) reported that dugongs are neither present in the Nicobar Islands (contrary to the report of Crusz 1960) nor in the Barren, Narcondom, Coco (Keeling), or Christmas Islands, although a population persists in the Andamans. Few are found in Burma, the Mergui Archipelago, or Malaya (Gibson-Hill 1950), and dugongs are reportedly extinct in Borneo and the Philippines (Philip and Fisher 1970; Wycherly 1969). Records exist from the Siamese coasts and the coasts of Sumatra (Allen 1942) but recent reports from these areas are lacking. The animals were said to be plentiful along the rockier parts of the Sarawak coast of Malaysia in 1932 (Gibson-Hall 1950), but Wycherly (1969) concluded that numbers have declined in that area, and only on the east coast of Sabah did dugongs still appear to be numerous. They are reported from the Moluccas although their present abundance there is unknown (Bertram and Bertram 1973).
Only one specimen has been recorded from Formosa (Hirasaka 1932), and a specimen was brought into Hong Kong in 1940 (Bertram and Bertram 1973). Dugongs were formerly abundant in the Ryu Kyu Archipelago (Hirasaka 1932), but they have since decreased and are today rare (Bertram and Bertram 1973; Fitter 1968; Allen 1942).
Records of dugong vertebrae, formerly used as arm rings by natives, suggest that these animals once occurred in the Pacific islands of the Lesser Sunda Group (Hirasaka 1934); however, no recent records exist from these localities. Presently, the range extends east to include the Palau Islands (Bertram and Bertram 1973; Harry 1956), the Carolines, New Britain, the Solomons, New Caledonia, New Hebrides, and Australia (Bertram and Bertram 1973; Philip and Fisher 1970; Jones 1967a). According to Rice and Scheffer (1968), dugongs are absent from the Marshall Islands (cf. Philip and Fisher 1970). They are reported from the Gilbert, Ellice, and Fiji Islands (Bertram and Bertram 1973).
Australia: Dugongs occur along the west coast of Australia as far south as Perth (Bertram and Bertram 1966b). Stable populations have been cited from Sharks Bay (Macmillan 1955; G. Heinsohn, personal communication) and Broome (Macmillan 1955). They are reportedly common all along the coast of the Northern Territory (Bertram and Bertram 1973), and especially at Barnambarnga and Melville Bay on the Cape Arnhem Peninsula, in the Cadell Strait between Echo Island and the mainland, in Popham Bay, and off Blackrock Point on opposite sides of the Cobourg Peninsula (Johnson 1964). In the Gulf of Carpentaria, dugongs are common near the mouths of the Rose and McArthur rivers, and are abundant around Mornington Island where aborigines take about 50 per year. They are also reported from the Mitchell River area and the Arukun Mission (Bertram and Bertram 1973).
Dugongs may be found along most coasts of New Guinea and they are common along the southern coasts of Papua (Bertram and Bertram 1973). In Torres Strait, the population appeared to be in good shape as of 1966 (Bertram and Bertram 1966a). However, islanders are still allowed to hunt them, and they are presently less common than earlier (G. Heinsohn, personal communication). Stable concentrations have been located off the east coast of Cape York, particularly in Prince Charlotte Bay (C. Barnett, personal communication). G. Heinsohn (personal communication) estimates the Townsville population to be 200-500, but adds that this is a very rough estimate since no attempt has been made to census the population.
Moreton Bay, Brisbane, once harbored large herds (Macmillan 1955; Troughton 1928; Anon. 1910), and commercial fisheries were locally established in the late 19th century (Bertram and Bertram 1966b). A population persists today, but numbers are so diminished that a commercial fishery could not be supported. Brisbane is now the southern limit of the range on the east coast of Australia, although dugongs were once found as far south as Botany Bay, Sydney, as evidenced by bones collected from middens of aborigines (Macmillan 1955; MacInnes 1951; Troughton 1928).
When the population density throughout the range is considered, dugongs are clearly most abundant in Australian waters. Present stocks there appear to be maintaining themselves and may even be increasing, especially in the northeast along the coast of Queensland (Bertram and Bertram 1966a and 1966b; G. Heinsohn, personal communication).
HABITAT PREFERENCE
The tropical or sub-tropical dugong is found in seas that range from 21-38 C throughout the year (Heinsohn 1972; Jonklass 1960). They do not appear to select a particular salinity, and while not known to ascend rivers [the single reference found referring to dugongs in coastal creeks (Jarman 1966) is questionable], they have been found in brackish coastal waters (Kingdon 1971; Hla Aung 1967; Jonklass 1960).
Four basic conditions necessary for proper dugong habitat are: (1) shallow saline water, (2) shelter from high winds and heavy waves, (3) an abundant food source, and (4) warm water. Unlike their estuarine relatives, the manatees, dugongs are totally marine and usually are found in coastal waters from 2 to 3 fathoms deep (Heinsohn 1972; Heinsohn and Birch 1972; Jarman 1966; Jonklass 1960). The sea floor at such depths is usually sandy with varying amounts of mud and silt, and ‘sea-grasses’ which form the major portion of the dugong diet grow here. These plants are intertidal and subtidal, generally occurring from the lower neap tide level to a depth of about 3 to 4 fathoms (Heinsohn 1972; Heinsohn and Birch 1972).
Dugongs are occasionally found at depths up to 8 fathoms, either feeding on deepwater plants, or escaping choppy waters at shallower depths (Kingdon 1971; Jarman 1966). Heavy winds and rough waters caused by seasonal monsoons, combined with exposure to coral reefs during rough weather, may cause appreciable mortality. In 1954, a large number of dead dugongs washed ashore following a cyclone on the south Indian coast (Jones 1967a), and Travis (1967) reported the death of a dugong on the Somali coast, apparently caused by heavy surf on the outer reef.
There appears to be some degree of regular daily movement within the general habitat. Dugongs reportedly spend most of the daylight hours in deep water. At dusk (Jarman 1966; Jonklass 1961), or with the rising tide (Kingdon 1971), they move to the shallower waters along established swimways thus avoiding the coral. On the basis of earlier reports, Annandale (1905) stated that large herds of dugongs once frequented the shallow waters of the Gulf of Mannar. Prater (1929a) assumed that the animals no longer inhabit the shallow water and that, whether singly or in herds, they remain in the deeper waters of the gulf. Because fishermen observed only sick or disabled dugongs along the shore, Prater concluded that there had been a shift in habitat preference from shallow to deep waters. No other such changes in habitat have been reported.
SEASONAL MOVEMENTS
Long distance migration is unknown for dugongs, but seasonal changes in their abundance in shallow coastal waters are apparent. These changes are correlated with the changing monsoon seasons and possibly with resulting shifts in abundance of food sources (Kingdon 1971).
Along the coast of east Africa, dugongs are most numerous in the shallow protected waters close to shore during the southeast monsoon (May-August). Exposed seas are then rough and winds are extreme. Sightings and catches are most numerous during this season (Kingdon 1971; Funaioli and Simonetta 1966; Jarman 1966). The dugongs may move to inshore waters to feed on Halophila, Zostera, and Diplanthera. These ‘grasses’ are virtually absent during the northeast monsoon (November-March) (Kingdon 1971). With the coming of the northeast monsoon, the animals travel to deeper waters farther from shore, and appear to be less concentrated. During this season, winds are gentle, and the waters calm (Kingdon 1971).
A similar movement, apparently in response to rough water, occurs along Indian coasts. Dugongs move inshore seeking shelter during the southwest monsoon (Jones 1967a). When dugongs were more numerous in the Red Sea, they were reported to appear annually on the Nubian coast near Aesa and to move north in January as far as Safadje (Anderson and De Winton 1902). Seasonal movements have also been reported in the Philippines (Seale 1915), but such movements are not evident along the coast of Queensland (G. Heinsohn, personal communication).
Dugongs are largely herbivorous and their historic range was broadly coincident with the tropical Indo-Pacific distribution of their food plants, ‘sea-grasses’ of the families Potomogetonaceae and Hydrocharitaceae (Kingdon 1971). Several food habit studies, conducted by analyzing stomach contents and observing feeding animals, indicate that dugongs are selective in their choice of food plants, and that this selectivity is probably based on palatibility and not on size and shape of plants.
Queensland: Heinsohn and Birch (1972) analyzed stomach contents of 15 animals taken off the coast of Queensland near Townsville, between May 1969 and April 1971. Contents were almost exclusively sea-grasses. Little sand, nonepiphytic algae, or silt was noted and, except for parasitic nematodes, no animal matter was found. Diplanthera uninervis and D. pinifolia were the most heavily used grasses, and D. uninervis was the most common species present in the area. Cymodocea serrulata, very common locally in subtidal and lowest intertidal flats off Townsville, was also an important food. Zostera and three species of Halophila were uncommon to rare in the stomachs. Zostera, commonly known as “dugong grass,” is locally abundant; however, extensive beds showed no signs of grazing by dugongs. These species are recorded as the major food items in New South Wales (Marlow 1962).
Except for Halophila, which occurs at depths from 6 to 9.5 m, sea-grasses are found growing on a sandy or muddy substrate within the reef from the lower neap tide level to depths of almost 6 m. Two abundant shallow water species, Syringodium isoetifolium and Enhalus acroroides, were not represented in the stomachs. Syringodium is within the size range of plants eaten, and is found at the usual feeding depths, but it may float too readily for easy feeding. E. acroroides has large, tough, fibrous leaves and stems, perhaps too tough for dugongs to graze. All regularly eaten plants have thin, relatively non-fibrous leaves and stems (Heinsohn and Birch 1972).
Sea-grasses are undoubtedly the primary food of dugongs, although a study of stomach contents from off Queensland indicated a marked change in diet during the year following the severe cyclone of 24 December 1971 (Spain and Heinsohn 1973). Twenty-nine individuals were examined in 1972, and algae, primarily the brown algae Sargassum (Phaeophycophyta) was an important component of the diet. Insignificant amounts of red algae (Rhodophycophyta) of the genera Laurencia, Spyridia, Hypnea, and Ceramium were also found. Sea-grasses were represented, but the amount of algae was substantial, and in three stomachs, little sea-grass was found.
A few colonial hydrozoans, parts of a holothurian, and an ascidian were also included among the contents. The cyclone probably caused considerable damage to the sea-grass beds by shifting substrate and by the large influx of fresh water, changing the salinity. When confronted with decreased amounts of preferred foods, the dugongs apparently began eating algae to supplement their diet. Further evidence of a shortage in food supplies is the marked increase in numbers of dugongs captured, which Heinsohn and Spain (1974) attribute to the influx of individuals from other areas searching for food.
Africa: Vast meadows of Syringodium and Cymodocea which occur off the coast of the Lamu Archipelago, Kenya, are the principal food plants in this area (Kingdon 1971). During the northeast monsoon, dugongs inhabit the deep water beyond the reef off Kiwaiyu, Kenya, where the animals feed on roots of Cymodocea rotunda (Jarman 1966). With the approach of the southeast monsoon, the dugongs move to sheltered bays, where they feed on Halophila ovalis and H. stipulacea, Zostera spp., and Diplanthera uninervis, plants which are largely absent during the northeast monsoon season (Kingdon 1971; Jarman 1966).
According to Petit (in Prater 1929a), dugongs in Madagascar subsist exclusively on Cymodocea australis, which is common in the local coastal waters. People in this area refer to C. australis as “dugong grass” (Prater 1929a).
Other Areas: In the Indian Ocean, dugongs feed largely on phanerogamous sea-grasses, i.e., Cymodocea (Jonklass 1961; Jones 1960; Prater 1929a). However, one specimen examined by Annandale (1905) contained large masses of green marine algae, possibly Caleurpa, according to Spain and Heinsohn (1973). Prater (1929a) explained this apparent change in diet over the years from algae to sea-grasses as a result of change in habitat preference. The animals were reportedly abundant in the shallows previously, but at the time of Prater’s report, few were seen along the shore. These differences may reflect changes in food availability, similar to the dietary shift noted in Australia (Spain and Heinsohn 1973).
In the Red Sea and the Malayan Archipelago dugongs eat phanerogamic sea-grasses (den Hartog 1970; Gohar 1957; Anderson and De Winton 1902). The stomach of a single specimen from Formosa contained marine algae and some crabs (Hirasaka 1932), probably one more example of an individual resorting to supplementary foods in the absence of the preferred ‘sea-grasses’.
BEHAVIOR
Feeding is the predominant activity of the dugong. The interlacing bristles on the lip pads are used for grasping sea-grasses (Gohar 1957; Prater 1929a), and the roughened facial disc is thought to be used in uprooting tubers and roots (Jarman 1966; Thomas 1966). Evidence for the rooting action by the face and mouth can be seen in the tusk wear of the males and in females possessing erupted tusks. The heavily worn and sometimes broken tusks suggest scraping and knocking against a hard substrate (Pocock 1940). Conspicuous feeding trails left through the sea-grass beds are composed of a series of circular patches, each about 30 cm wide and 60 cm long, and follow a slightly wandering course. These paths, often following the contour of a sand bank, are completely cleared of vegetation, exposing the substrate and providing further proof that the shovel-face is used for digging.
Gohar (1957), on the basis of callouses found on the edges of the flippers, hypothesized that the forearms were used to dig roots. Subsequent observations do not support this hypothesis (Jones 1960; C. Barnett, personal communication). While grazing, dugongs move along the bottom by “walking” on their flippers (Jarman, 1966) as do Florida Manatees (Hartman 1971). Dugongs have also been observed drifting with the flippers dragging against the substrate, which may create the calloused areas (C. Barnett, personal communication).
A captive dugong was observed stuffing cut grasses into its mouth with its flippers (Jonklass 1961), demonstrating the flexibility and control of forelimbs. After grasping each mouthful of food, the animal shook its head back and forth in an apparent effort to remove mud or sand. This action is apparently effective, for analyses of stomach contents show very little of these materials (Heinsohn and Birch 1972; Prater 1929a).
Jarman (1966) reported that dugongs favor night feeding and spend daylight hours in deeper offshore waters. Kingdon (1971) contends that tides are even more important than light in determining daily movements. He concludes that dugongs prefer to feed with the rising tide, and that they follow the tide out with the ebb. C. Barnett (personal communication) observed dugongs off the coast of northeast Australia coming inshore most often on a rising tide, whether at dawn or dusk, although most commonly on overcast days.
Dugongs generally remain submerged from 11/2 to 4 min (Jones 1967b). In 108 timed dives of a 1.8 m female dugong, the average submergence time was 3:01 (range 0:33-8:26) (Kenny 1967). Time between opening and closing the nostril flaps is about 12 s (Jones 1967b) and the interval between surfacing and submerging is from 2 to 6 s [seconds] (Kenny 1967). Traveling dugongs reportedly rise to breathe less frequently than do feeding animals (Jarman 1966).
When swimming to and from feeding grounds, dugongs travel at about 10 km/h (Jarman 1966), but when disturbed, they are capable of greater speeds. Jonklass (1961) estimated a maximum speed of 18.5 km/h over short distances.
The only grooming behavior thus far observed is that of rolling and rubbing the body in the sand (C. Barnett, personal communication; Jonklass 1961), possibly an attempt to relieve itching. Defecation occurs frequently with some flatus and this sign (rising bubbles) has been used by native hunters to locate their prey.
Vocalizations from animals in distress are described as whistling sounds (Kingdon 1971). Calves are said to have a bleating lamb-like cry (Troughton 1947). These sounds apparently are without a high frequency component and are believed to be used only for short range communication (Kingdon 1971).
Dugongs formerly occurred in large herds of up to several hundred animals (Stoddart 1972; Bertram and Bertram 1966b; Annandale 1905). Although large herds are now a rarity, Travis (1967) reported a large herd of about 500 off the open coast of Somali. Dugongs swam both within and beyond the reef and were neither elusive nor shy. The young calves left the herd in the afternoon to form a sort of nursery close to the sandy beaches, and when Travis waded into the water, the calves swam about his legs, often rubbing against them.
Dugongs were also reported feeding in herds off the coast of Broome, Australia, in 1928 (Prater 1929a). Today, however, dugongs are most commonly found singly or in small groups of up to six individuals (Hughes and Oxley-Oxland 1971; Kingdon 1971; Jarman 1966; Troughton 1947; Prater 1929a). C. Bertram (personal communication) suspects that unwary herding behavior was natural for the species before they were massively exploited and that the surreptitious behavior of most dugongs today is a direct result of hunting pressure.
Sex and age composition of dugong groups vary. Calves usually accompany their mothers for a period of over a year, forming a stable social unit for that time (Macmillan 1955); males are not thought to remain with this unit (Jarman 1966). Several small groups may converge to feed and associate within the same area, but social interactions under these circumstances are unknown.
In Manda Bay, Kenya, associations of over 80 individuals have formed in recent years. These occur most frequently during the peak of the hot season (Jarman 1966). Such concentrations may form in response to a concentrated food source, or a synchronized estrous in several cows may attract large numbers of males. Stimuli responsible for such concentrations of dugongs are unknown.
Dugongs are thought not to be especially intelligent. The brain is small and has few convolutions (Gohar 1957; Dexler 1912a). A pair of dugongs kept in captivity for 62 years exhibited no noticeable change in behavior and never appeared to recognize their feeders (Jones 1967a). The animals tame rather quickly, and this is taken by some investigators as evidence of some degree of intelligence (Jonklass 1961; Troughton 1947). Rarely have dugongs been observed at play (Jonklass 1961). A degree of sociability was indicated when one of a pair was captured and the other lingered about the area, apparently looking for its companion (Troughton 1947). Likewise, Jones (1967a) observed the male of the captive pair at Mandapam Camp, India, make vigorous attempts to prevent separation from the female. To date, neither fighting nor aggressive behavior has been recorded. . . . . . .
EXPLOITATION
Throughout history man has considered the dugong as a product, existing for his personal exploitation. Although dugongs have numerous uses (meat, oil, leather, etc.), their aesthetic qualities, of being interesting and specialized mammals, have been neglected.
The flesh of dugongs, likened to tender veal or pork, has long been sought by man. It also has the unusual and thus far unstudied property of “keeping” for a considerably longer time than other meats without spoiling, even at high temperatures (Annandale 1905; G. Heinsohn, personal communication). When rolled and smoked, it is similar to bacon (Troughton 1928).
An average-sized adult yields from 19 to 30 liters of oil which is similar to cod liver oil. The thick hide tans to a good grade of leather especially well suited for sandal-making. Dense bones and the tusks provide a source of ivory for carving, or they can be prepared as an excellent charcoal used in sugar refining (Troughton 1928).
Throughout the range of the dugong, different cultures believed that products of the dugong possess medicinal and aphrodisiac properties. As early as 1665 the Chinese believed the tympanic bones, when ground to a fine powder, would clear the kidney of its afflictions (Allen 1942). In Malaysia, the tears of landed beasts were collected for use as an aphrodisiac. The fishermen of Ceylon regularly ate the meat, believing it had a rejuvenating and aphrodisiac quality (Jones 1967a). In the Aragusuku Islands of the Pacific, dugongs were hunted in a yearly tribute to feudal lords (Hirasaka 1934).
Today, although many of these superstitions have been discarded, the dugong still provides a palatable source of protein. In Kenya and the Republic of Somali, the traditional method of hunting the dugong is with a spear after luring the animals to a bait of cut grass. Nets are also used, but accidental captures in shark nets probably outnumber those actually killed during purposeful hunting (Jarman 1966). In Brava, Somali, only one or two animals are netted each year (Funaioli and Simonetta 1966).
Although vigorously hunted there in the past, the dugong now ap- pears fairly secure from hunting pressure in Tanzania and on Mafia Island, where fishermen seldom take them (Savory 1958). Hughes and Oxley-Oxland (1971) did not find hunting pressures great in the Antonio Enes area of Mozambique. The single fisherman who attempted to catch dugongs there considered it a good month if he got two animals. Arab fishermen in the Red Sea once prized the teeth (Allen 1942), but population numbers are now so low that there is no longer deliberate hunting for them (Gohar 1957).
In the Andaman Islands, natives netted extensively for dugongs in the past but, since the Andaman Islanders are now practically extinct, it is probable that the remaining dugongs are relatively safe (Prater 1929a). Previously, fishing for dugongs in the Gulf of Mannar and in Palk Bay, Ceylon, was intense. The meat was especially desired by the Moslems who used it as a pork substitute (Crusz 1960; Prater 1929a). Following the establishment of train transportation between Mannar and Colombo, on the southwest coast of Ceylon, the prices of dugong meat soared, and large numbers of dugongs were shipped alive to market (Phillips 1927). A commercial fishery for dugongs also operated in the Gulf for some time, but decreased supplies of dugongs forced them to close (Prater 1965).
Despite protective legislation in Ceylon, there is still a ready market in Jaffna, Colombo, and Negombo; however, it appears that concerted netting of dugongs has come to an end. Accidental captures probably have a considerable effect on the population, and dugongs reach the market in Ceylon at a rate of about 100 per yr [year] but this rate is now dropping (Bertram and Bertram 1970a). The increasing rarity of the dugong is attributable to intensification of the marine fisheries in the area (Bertram and Bertram 1970a).
In the western Pacific, north of Australia, dugongs were exposed to heavy hunting pressure during World War II, especially in the Palau Islands where they were almost extirpated (Harry 1956). No record of more recent exploitation in these islands has been found.
Although aborigines of Australia have long hunted dugongs (Troughton 1928), populations were maintained at high densities until the white man began hunting and netting on a large scale (Anon. 1910; Fairholme 1856). Today, hunting is restricted to aborigines and Torres Islanders, thus heavy pressure has been lifted from the Australian population. Shark nets do, however, present a problem to Australian dugongs. The government-sponsored program of netting the beach areas for sharks has resulted in large scale drownings of dugongs in Queensland (Heinsohn 1972). In the vicinity of Townsville alone, 82 dugongs were caught the first year, and between 6 and 16 for each of the 6 yr [years] following. These efforts to control sharks seem to have decimated the local Townsville population (Heinsohn 1972).
It has since been decided not to extend shark netting into new areas (G. Heinsohn, personal communication), and the Queensland government will resort to alternative methods to protect humans from sharks.
LEGISLATION AND PROTECTION
Several African countries have enacted protective legislation for the dugongs (Table 2). [Not included.] While protection seems nearly complete, problems of law enforcement and education remain (Hoffman and Jungius 1972). In Kenya, enforcement appears to be effective and dugongs are not deliberately hunted (Bertram and Bertram 1973). Some hunting is, however, still carried on in Mozambique, despite protective legislation (Hughes and Oxley- Oxland 1971). Dugongs are protected in the recently proclaimed Paradise Islands National Park in Mozambique (Hughes 1973), but that park seems to be the only one in Africa where dugongs are found. The government of Kenya plans to establish a terrestrial-marine national park in the Lamu region, where apparently there is a stable dugong population. The Kenya government also intends to open the new Kasiti Marine National Park in the Shimoni area (Anon. 1973) where the coast line also supports dugongs.
Following a survey of the Tanzanian coast for suitable sites for marine parks and reserves, Ray (1968) recommended at least two areas in which dugongs are known to reside. These are the Mafia Island-Rufigi Delta and the coastal area of Kiliwa. Although no further progress has been made toward the actual establishment of these reserves, Tanzania is now working for more conservation- and tourism-oriented programs (Hoffman and Jungius 1972; Ray 1968) which may include development of these parks in the future.
As early as 1928, dugongs were noticeably decreasing in numbers in India and Ceylon, and recommendations were made for their protection (Phillips 1929; Prater 1929a). No action was taken, and as recently as 1960 the dugong was still on the “huntable” list of the Department of Fisheries of Ceylon. At that time, the Department of Wild Life wanted to place the animal on their protected list, but lacked an adequate staff to provide enforcement (Jonklass 1960). Sanctuaries were suggested as a preferable alternative and sites along the Wilpattu Reserve coast line or in Puttalam Lagoon were proposed (Fitter 1968; Jonklass 1961; Spittel 1960), but as of 1971, no parks or reserves in either Ceylon or India contained dugongs (Ross-Macdonald 1971).
Protective legislation was finally passed by the government of Ceylon in 1970, and India followed suit in 1972. Although fishing for these animals has ceased in these waters, accidental nettings are increasing due to a growing marine fishery. It has been suggested that some areas, such as Dutch and Portugal Bay or Puttalam Lagoon, Ceylon, be placed off limits to fishermen (Bertram and Bertram 1970a). To date, this has not been done.
Existent legislative protection for dugongs in the Indian Ocean and Pacific islands is extensive (Table 2). [Not included.] All hunting of dugongs within Australia was banned in March 1969 (Anon. 1969), except by aboriginal populations which hunt them regularly in the Gulf of Carpentaria (Bertram and Bertram 1973).
SUGGESTIONS FOR CONSERVATION
Basic research based on well-designed field work is desperately needed for this species. Although this report presents much of the available information, it must be remembered that these bits and pieces were collected from a large number of sources and cover a time span of over 100 years. Most of the literature contains second-hand or anecdotal information, and it is difficult to ascertain the reliability of such information.
Comprehensive research along two separate lines could provide the answers to many of the remaining questions. First, by marking animals, we could for the first time obtain numerical data on dugong populations. Thus far, population estimates have been restricted to such terms as: declining, stable, common, etc., and a more systematic method of censusing clearly is needed. Movements of marked individuals could then be recorded and monitored, especially by follow-up aerial surveys. Marked animals would also provide a means of collecting demographic data on populations.
The second approach would include underwater observation of dugongs in their natural habitat, such as has been done on the West Indian Manatee in Florida (Hartman 1971). Such a study could define the ecological requirements for maintaining stable dugong populations, and assess the impact of the dugong upon its own environment. Observations of free ranging dugongs may also yield information concerning all aspects of dugong behavior and reproductive biology. The feasibility of such a project is uncertain because suitable study areas with calm, clear water and reliable numbers of dugongs may not exist. Possible localities are: Antonio Enes, Mozambique; the Lamu-Malindi area, Kenya; Cape York Peninsula, Arnhem Peninsula, or Sharks Bay, Australia.
Although no quantitative assessment has yet been made of the damage man inflicts upon the dugong’s environment, it is almost certain that increasing human population densities impose a substantial threat to this species. Dugongs are extremely shy and are known to flee from men and boats. Furthermore, man’s pollution of offshore marine habitats assuredly affects the food supply and quality of the water in which these creatures live.
Marine parks, where protection is maximized, are highly recommended for conservation of this species. The need is greatest in the Indian Ocean, along the shores of India and Ceylon, to provide sanctuary for dugongs from the marine fisheries. Certain individuals have for some time stressed the concept of dugong reserves in this area without results. The establishment of marine parks, with personnel concentrated along coastal areas, would provide more effective protection from poachers, fishermen, and shark netters throughout the range of the dugong.
Sandra L. Husar, A Review of the Literature of the Dugong (Washington, DC: Fish and Wildlife Service, 1975). pp. 1, 9-17, and 21-25. Dugong map appears in this cited work on page 9. Drawing appears on page 531 in Allen, Extinct and Vanishing Mammals of the Western Hemisphere. Black and white images appear on pages 42, 126, and 127 in E.J. Stuart, A Land of Opportunities: Being an Account of the Author’s Recent Expedition to Explore the Northern Territories of Australia (London: John Lane, 1923).
Color images are from: https://en.wikipedia.org/wiki/Dugong
See: https://babel.hathitrust.org/cgi/pt?id=uc1.31822012535860&seq=1